Risk management and instructions for use
Risk management plays a central role in assessing the safety and performance of your medical devices. Risk analysis integrates the results of clinical evaluation and post-market surveillance (PMS). In the event of anomalies in PMS or an incident report with subordinate measures, risk management determines the further procedure. Risk analysis as a critical element of risk management and the instructions for use as part of risk control constitute a main part of your technical documentation.
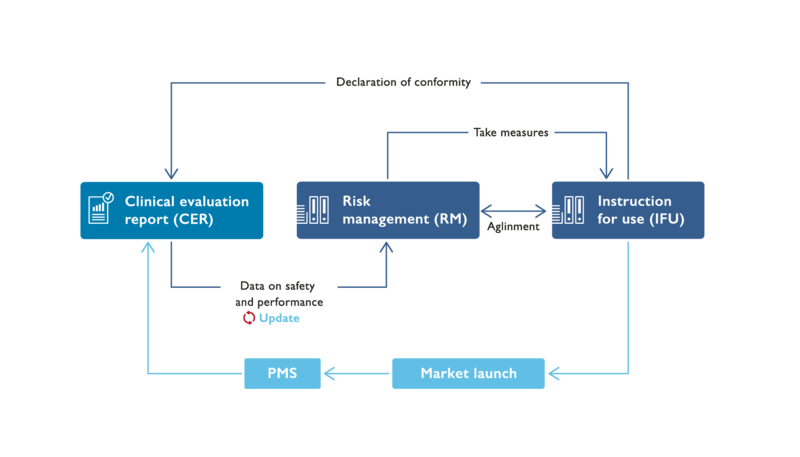
Risk analysis and instructions for use must meet various levels of normative requirements, including ISO/EN ISO 14971 as well as the higher level MDR (Medical Device Regulations (EU) 2017/745). Other interfaces to other documents include clinical evaluation, post-market surveillance (PMS), biological evaluation and usability.
Risk analysis, instructions for use and novineon CRO: your benefits
We have a neutral, objective view of your risk analysis and instructions for use and can recognize possible inconsistencies or inaccuracies with respect to content. We advise you on methodological and clinical issues in order to jointly define a suitable risk management strategy and instructions for use for your medical devices. Based on the requirements of current regulations and standards, we review, revise or write your documents so that they are plausible, permanently traceable and easy to update.
Our many years of experience enable us to advise and support you competently, reliably and individually: from the formal review of the instructions for use and/or risk management file, through workshops on individual topics, to the development of the instructions for use/risk analysis.

