Quantitative clinical evaluation qCE
The stricter requirements that MDR places on clinical evaluation increase its complexity. Especially for medical devices that have been on the market for a long time, as well as for class III medical devices, where the clinical evaluation must be updated at freuent intervals, the scope of the clinical evaluation increases considerably due to the constantly increasing amount of clinical data.
To the extent that the data structure of published studies permits (comparable endpoints, comparable study designs, etc.), our quantitative clinical evaluation statistically summarizes the relevant safety and performance parameters and presents them in graphic form.
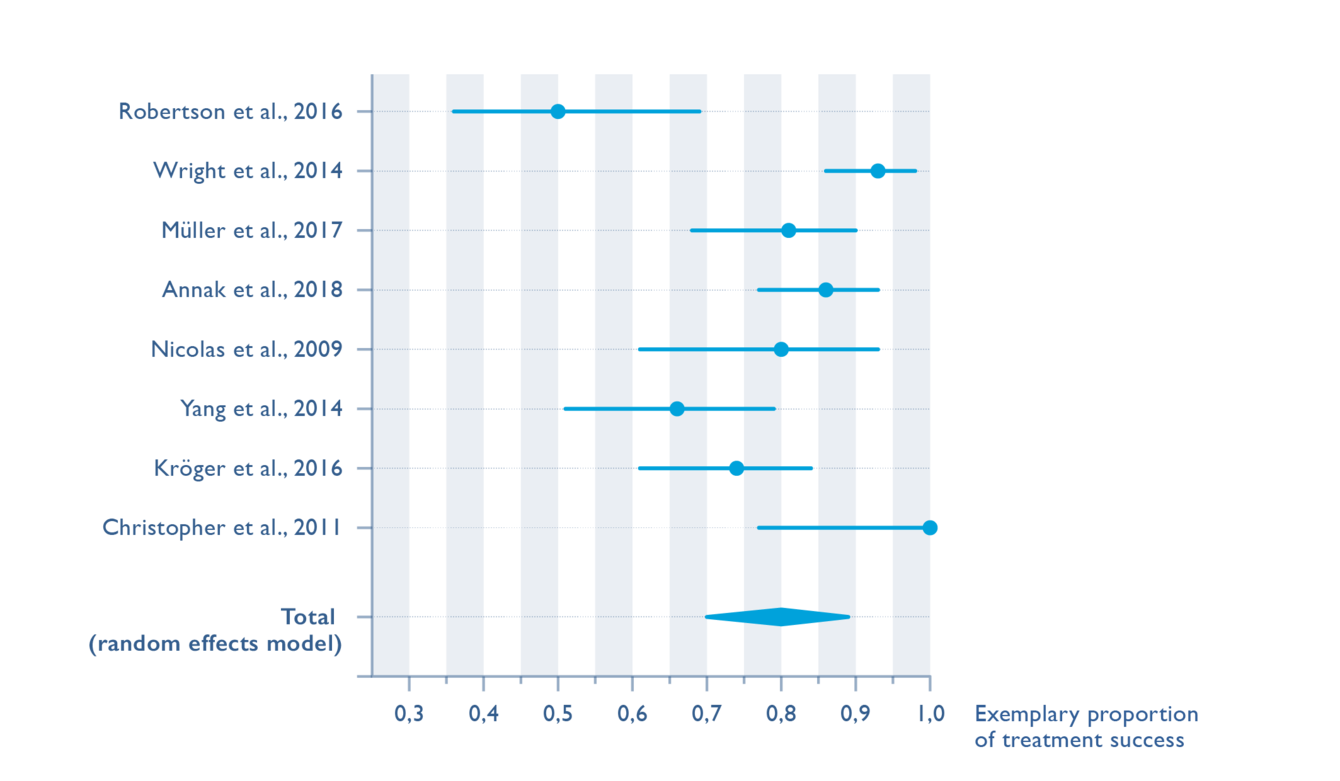
The quantitative clinical evaluation concentrates on the state-of-the-art in procedures and devices that have been clinically researched extensively and presents the collected clinical data in a structured analysis. The summarized presentation facilitates the writing of further reports such as the Summary of Safety and Clinical Performance and can support scientific marketing.

