Performance evaluation
Performance evaluation is an essential part of the life cycle of an in vitro diagnostic medical device (IVD). The purpose of such an evaluation is to align the essential safety and performance requirements. To this end, data on scientific validity, analytical performance and clinical performance are assessed to provide clinical evidence for an IVD.
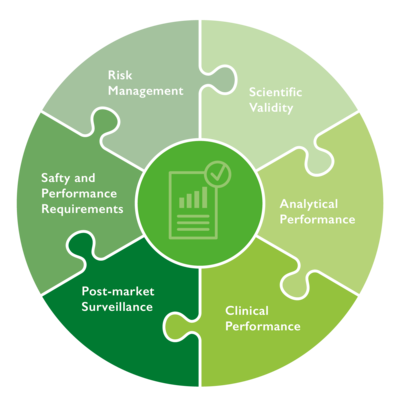
Risk management serves to identify and evaluate risks. In this way, risk management is closely linked to performance evaluation; they should thus be carefully coordinated.
In order to continuously monitor the safety and performance of an IVD after market launch, information from post-market surveillance (PMS) and post-market performance follow-up (PMPF) is included in the performance evaluation. This ongoing process is described in a performance evaluation plan.
novineon CRO brings structure to the planning and execution of your performance evaluation. Together with you, we create a plan and report for the performance evaluation of your IVD. Optionally, we can manage various aspects of the performance evaluation for you, e.g. a systematic literature review for the report on scientific validity and clinical performance.

