In the European market, different regulatory requirements apply depending on the product group. For example, food safety is subject to Articles 168 and 169 of the Treaty on the Functioning of the EU, food supplements to Directive 2002/46/EC and medicinal products to Directive 2001/83/EC. Difficulties are caused by product groups that cannot be clearly assigned to one regulation or that fulfill several regulatory definitions. Such products are called borderline products. So how do you deal with a product like a massage table, which can be used both for the treatment of severe tension and for a relaxing massage after the gym? In this article we describe the definitions of medical devices, medicinal products as well as combination and wellness products.
Definition of medical device according to Article 2 (1), MDR
Medical devices are products that, according to the manufacturer, are intended to be used in human beings for one or more of the following medical purposes:
- diagnosis, prevention, monitoring, prediction, prognosis, treatment or alleviation of disease,
- diagnosis, monitoring, treatment, alleviation of, or compensation for an injury or disability,
- investigation, replacement of modification of the anatomy or of a physiological or pathological process or state,
- providing information by means of in vitro examination of specimens derived from the human body, including organ, blood and tissue donations,
- and which does not achieve its principal intended action by pharmacological, immunological or metabolic means.
Explicitly excluded from the MDR are, among others, cosmetics, in vitro diagnostics and medicinal products.
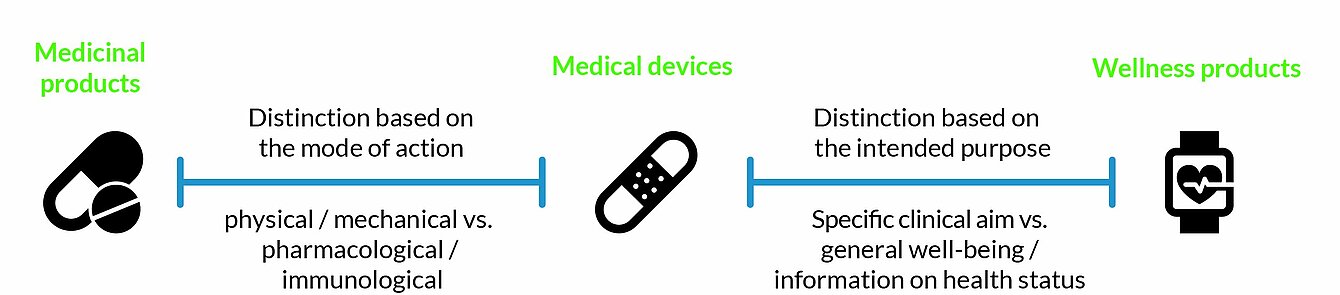
Thus, the definition of a medical device is divided into two parts, which must be fulfilled for the product to be classified as a medical device: First, the intended purpose must be medical; and secondly the desired effect must be based on the physical or mechanical properties of the product. There are a few exceptions to this rule, including (colored) contact lenses or liposuction devices, which are listed in the MDR under Annex XVI.
Differentiation between medical devices and wellness and cosmetic products
The distinction between wellness products and medical devices is based on the intended purpose. Thus, technically identical products can be marketed as medical devices or as wellness products.
There are many examples of such borderline products:
- Personal scales for diagnostic purposes are classified as medical devices. For example, a medical scale with bio-impedance analysis could be used to diagnose muscle loss or obesity. The same scale could output muscle or body fat percentage data without a diagnostic context, making it a bathroom scale for home use.
- Exercise equipment can be used for sports as well as for medical purposes such as rehabilitation or physical therapy. Again, the wording of the intended purpose in the instructions for use determines whether a product is a medical device according to the MDR.
- The massage products already mentioned above count - if pain reduction or improved blood circulation are claimed - as medical devices. Pure relaxation products belong to the wellness sector.
Important
If a product is to be marketed as a wellness product, no clinical performance may be claimed or disease referenced. In addition, wellness products are intended to be used only by healthy people, not sick people. The following advertising claims may be considered "non-medical":
- feel or look healthier,
- improve general health,
- measure fitness or performance (such as pulse, number of steps, distance traveled, etc.),
- improve (personal) hygiene,
- promote a healthy routine.
Cosmetics are similar to wellness products, as long as the product does not fall under the definition of medicinal products (see following chapter). A product can be considered a cosmetic or a medical device, depending on its intended effect (healing and relief versus improving hygiene, etc.). In fact, the ingredients of cosmetics are currently more regulated than those of medical devices, and the list of banned cosmetic substances is not applicable to medical devices. Thus, potential contact allergens or denatured alcohol, among others, can be used in medical devices, even if they have long been banned in cosmetics.
Distinction from medicinal products
Pharmaceuticals are among the most strictly controlled products. According to the Association of Research-Based Pharmaceutical Companies in Germany, the average approval of a new drug takes about 13 years and costs about $1.0 - $1.6 billion.
Pharmaceuticals are defined on the basis of Directive 2001/83/EC:
- Any substance or combination of substances presented for treating or preventing disease in human beings
- Any substance or combination of substances which may be administered to human beings with a view to making a medical diagnosis or to restoring, correcting or modifying physiological functions in human beings is likewise considered a medicinal product.
From the first subpoint of the definition alone, medical devices and medicinal products cannot necessarily be distinguished, as both are used to alleviate, treat, or prevent pathological conditions. Thus, medical devices are distinguished from medicinal products by the second subpoint of Directive 2001/83/EC. According to MDCG 2022-5, the decisive factor in individual cases is the "mode-of-action", i.e. the principle of action. Products whose main principal of action is physical or mechanical are considered medical devices. Substances that act metabolically, immunologically or pharmacologically are medicinal products. The principle of action is determined objectively on the basis of the state-of-the-art in the scientific literature.
In case of doubt to which group a certain product belongs, it is classified as a medicinal product in accordance with Article 2(2) of Directive 2001/83/EC.
Combination products
Medical devices can contain medicinal products in order to support their effect. This results in combination products, which consist of a medical device and a medicinal product, but whose individual components would be one or the other. This product category has to be considered separately from a regulatory point of view, because a combination product needs, depending on its main mode of action, an certification under MDR with additional files according to Directive 2001/83/EC or an certification under Directive 2001/83/EC with additional files according to MDR. In addition, not every product containing a drug substance is automatically a combination product. Therefore, a separate article on this topic will be published shortly.
Conclusion
- The definition of a medical device is divided into two parts: firstly, the intended purpose must be medical and secondly, the desired effect must be based on the physical or mechanical properties of the product.
- The differentiation between medical devices and medicinal products usually takes place on the basis of the main mode of action.
- The main mode of action is determined on the basis of the state-of-the-art in the scientific literature.
- In case of doubt, a borderline product is classified as a medicinal product in accordance with Article 2(2) of Directive 2001/83/EC.
- The distinction from wellness products is based on the intended purpose defined by the manufacturer for the product.
- If marketing of a device as a wellness product is desired, no clinical performance may be claimed, or disease referenced.
Dominik Heiss, B.Sc.
Consultant
dominik.heiss@novineon.com
+49 7071 / 98979 - 150



